  
In fact, sulfuric acid is one of the seven strong acids. This places the acid in higher rank compared to other acids. Sulfuric acid has a Ka value of 1.0 x 10 2. The larger the Ka, the stronger the acid as its ions dissociate more completely in a solution. This is directly associated with the ratio between the formation of hydronium ions and the molecular acid. The acid dissociation constant is also known as the chemical reaction equilibrium of acid with its base conjugate. The pH may change based on the concentration but not the Ka. This constant, represented by the symbol Ka, does not change regardless of the concentration of the acid. Just like other acids, the strength of sulfuric acid is measured by its acid dissociation constant in a solution. This means that each oxygen is bonded at an angle of 109.5 degrees from the other oxygen atoms. The other two atoms are single-bonded with sulfur but have hydrogen attachments.īecause of valence shell electron pair repulsion (VSEPR), a molecular model that predicts the geometry of the atoms, the oxygen atoms spread out in tetrahedral form. Two of the oxygen atoms are double-bonded with the sulfur. Structurally, a molecule of sulfuric acid, which has a molecular weight of 98.079 g/mol, has a central sulfur atom that is surrounded by four atoms of oxygen. Sulfur to make sulfuric acid stored in a factory Sulfuric acid structure In the third stage, a catalyst known as vanadium (V) oxide on silica is used to convert sulfur dioxide to sulfur trioxide at 700 K.įinally, the sulfur trioxide is converted to sulfuric acid by allowing it to react with water at a constant temperature of 400 K. At this stage, the sulfur is burnt, converting it to sulfur dioxide. Molten sulfur is then blasted by dry air at 1300 K. As shown in the first equation above, sulfur can also be extracted from metal ores as sulphides. 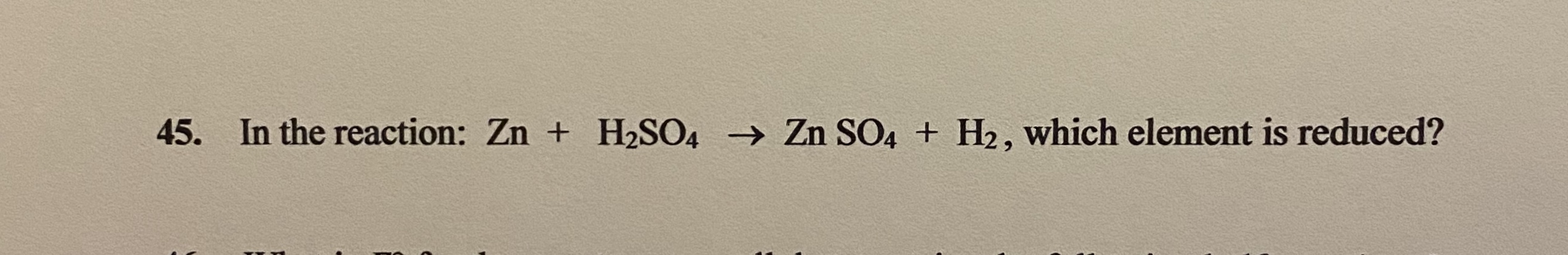
Unrefined petroleum and natural gas contain sulfur compounds that are removed during the refining process. Typically, sulfur is extracted from fossil fuels. 4 th stage – conversion of sulfur trioxide to sulfuric acid – SO 3 (g) + H 2 O(I) ? H 2 SO 4 (I).3 rd stage – conversion of sulfur dioxide to sulfur trioxide – 2SO 2 (g) + O 2 (g) ? 2SO 3 (g).2 nd stage – conversion of sulfur to sulfur dioxide – S(s) + O 2 (g) ? SO 2 (g).1 st stage – extraction of sulfur – 2PbS(s) + 3O 2 (g) ? 2PbO(s) + 2SO 2 (g).These stages are represented by various chemical reactions: Basically, the manufacturing process of this acid involves four stages. To understand this equation, you first need to know how it is manufactured. What colour does sulfuric acid turn blue litmus paper?.What is the equation for sulfuric acid?. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
